What We Do
Mechatronics design


Industrialization
We offer our clients a turnkey project management solution: after the approved design phase, we handle the entire manufacturing management process. Through comprehensive inspections and risk identification, we continuously improve our products’ characteristics, the productivity of our manufacturing, and overall quality. We hold ISO9001, ISO13485, and ISO14001 certifications, and we are familiar with the requirements of industry-specific documentation management. We support our clients with quality tools such as APQP, statistical process control, capability analyses, measurement system analyses, gauge repeatability and reproducibility studies, etc. Through our integrated ERP system, we are also able to provide our customers with manufacturer compliance and data analysis documents (CoC, CoA).










Manufacturing
- Our parts manufacturing covers a wide range:
- Up to 5-axis turning and milling
- Gear milling
- Wide range of sheet metal processing
- Steel and aluminum spot and arc welding
- Lead processing
- Gluing
- Plastic coverings
- Solvent-based painting
- Assembly and testing of cables
- 3D printing (FDM)





Integration and testing
Complete modules, consisting of various mechanical and mechatronic components, cables, and electronics, are assembled by our colleagues based on carefully developed and strictly controlled operational instructions. The assembly and integration are supported by our own designed, optimized tools, devices, and functional test equipment. Full traceability is a basic requirement for us. We track all manufacturing data of each component from receipt, through the production processes, all the way to the finished product. This ensures a safe and responsible manufacturing process for us.

Supply chain management
We manage and optimize the manufacturing process through multi-continent parts procurement. We take care of selecting and evaluating supplier partners, procurement, inventory management, and delivering finished products anywhere in the world, including lifetime spare parts supply. Upon request, we also provide customer-specific logistics solutions, such as supplier-managed inventory (consignment warehouse management).
We work with more than 600 supplier partners from all over the world. Our clients can even pre-select suppliers based on their own preferences. We choose our supplier base based on the necessary capabilities, quality, costs, and trust. With our supplier partners, we strive to create a win-win situation. One of the key factors of our success and part of our company philosophy is close collaboration with our suppliers.

Life cycle management
At Mediagnost, lifecycle management begins with functional test models and ends with the expiration of the product’s service obligation. To carry out these activities, we have established the appropriate processes, supported by comprehensive change management and an integrated enterprise management system. It does not matter if certain components or manufacturing technologies are no longer available later on: we offer optimal solutions to our customers to ensure that their products remain competitive for many years.
Our References
Philips Medical Systems
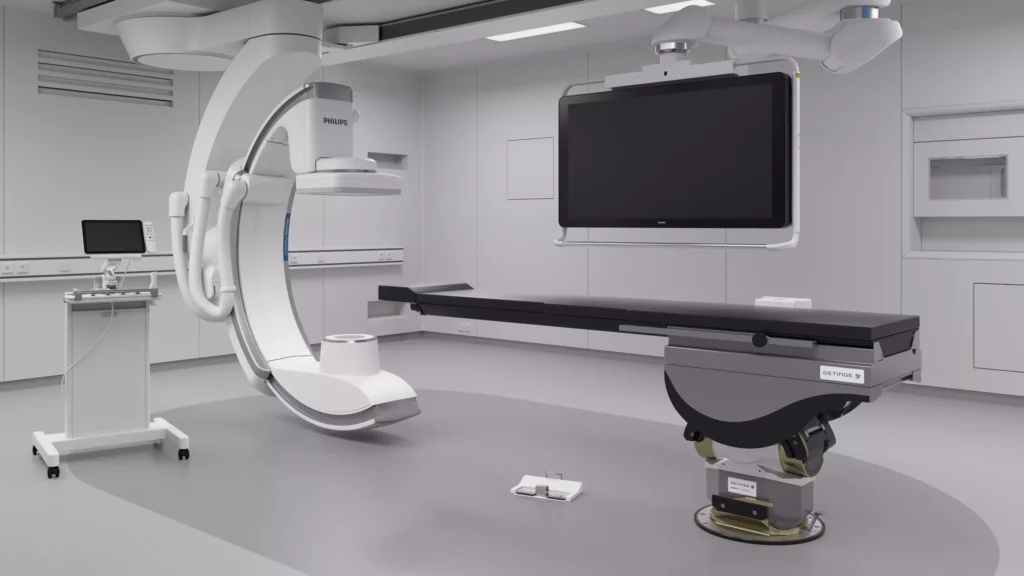
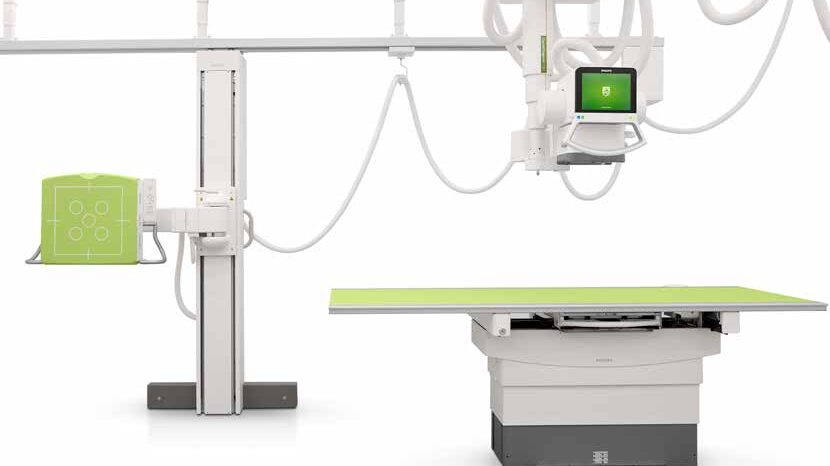
Philips DXR
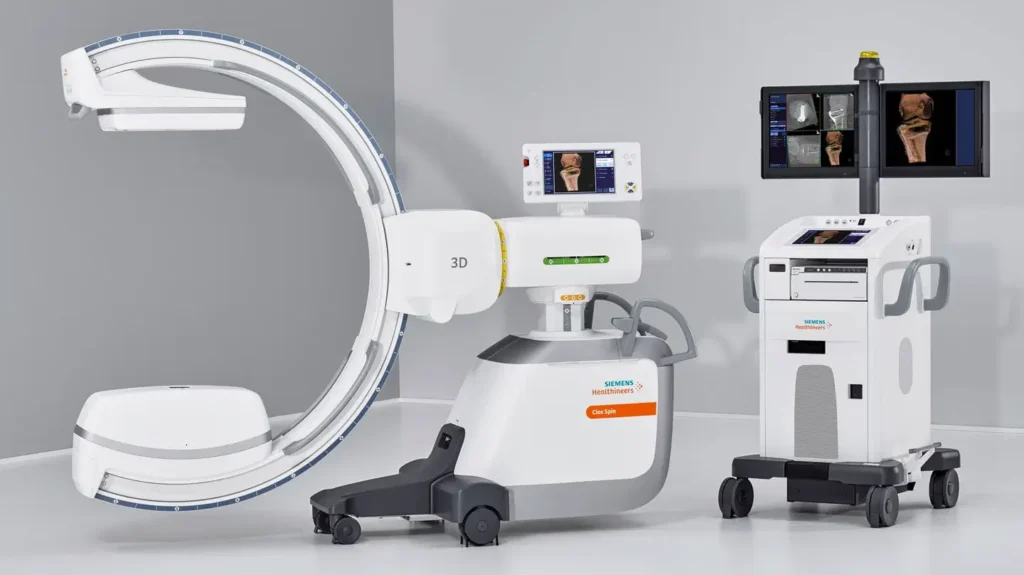
Siemens Healthineers
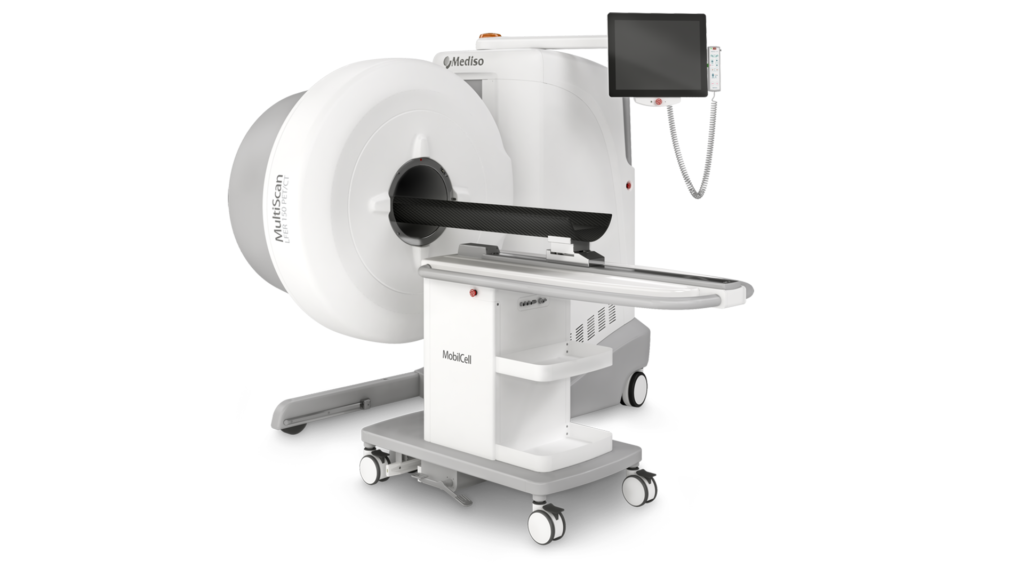
Mediso
Control-X
Innomed Medical
